 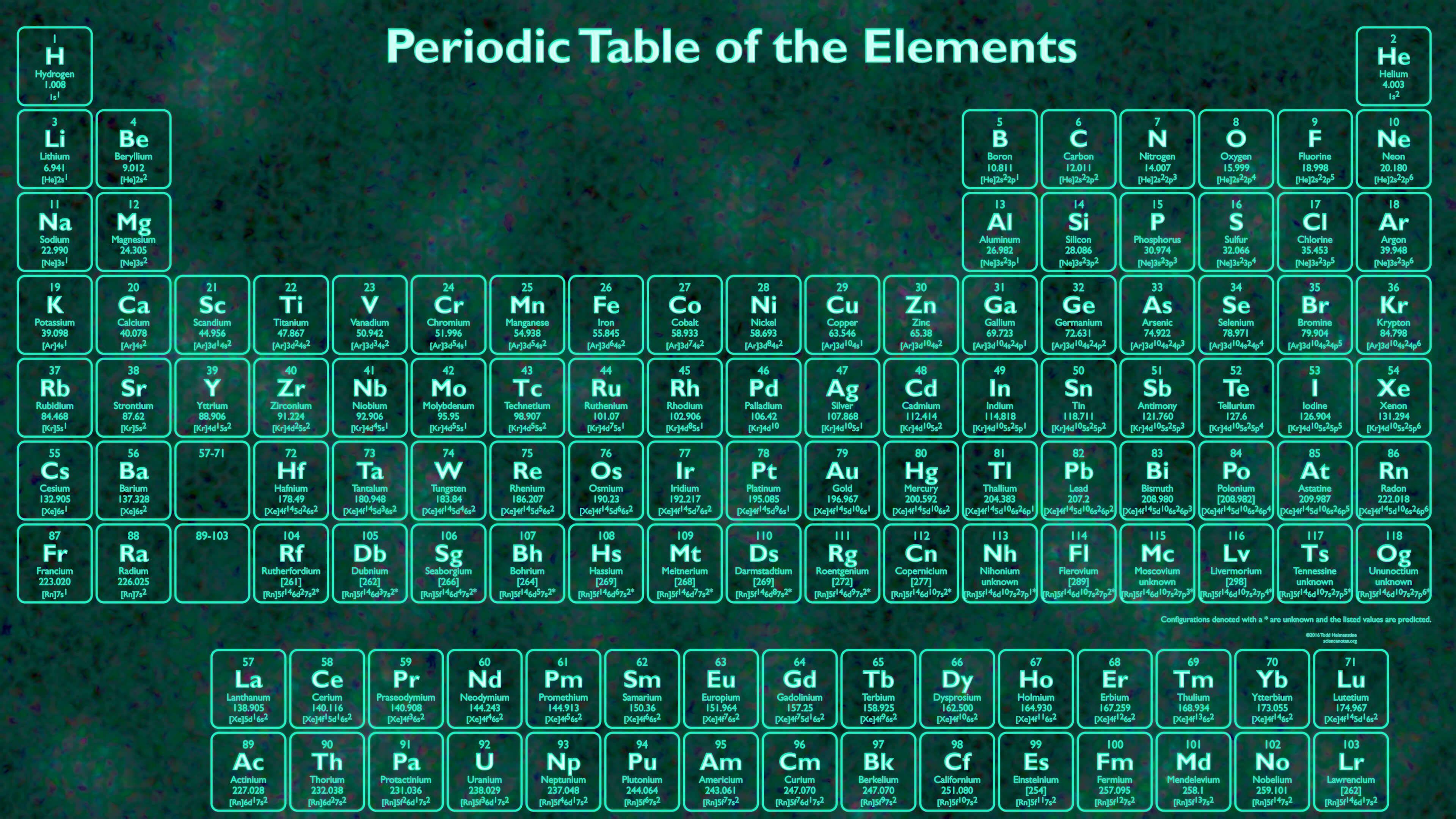 
Pekka Pyykkö and Burkhard Fricke used computer modeling to calculate the positions of elements up to Z = 172, and found that several were displaced from the Madelung rule. Models that take relativistic effects into account predict that the pattern will be broken. Seaborg's version of the extended period had the heavier elements following the pattern set by lighter elements, as it did not take into account relativistic effects. Īccording to the orbital approximation in quantum mechanical descriptions of atomic structure, the g-block would correspond to elements with partially filled g-orbitals, but spin–orbit coupling effects reduce the validity of the orbital approximation substantially for elements of high atomic number. Despite many searches, no elements in this region have been synthesized or discovered in nature. The first element of the g-block may have atomic number 121, and thus would have the systematic name unbiunium. An eight-period table containing this block was suggested by Glenn T. Any additional periods are expected to contain a larger number of elements than the seventh period, as they are calculated to have an additional so-called g-block, containing at least 18 elements with partially filled g- orbitals in each period. All elements in the eighth period and beyond thus remain purely hypothetical.Įlements beyond 118 will be placed in additional periods when discovered, laid out (as with the existing periods) to illustrate periodically recurring trends in the properties of the elements concerned. As of July 2023, the element with the highest atomic number known is oganesson ( Z = 118), which completes the seventh period (row) in the periodic table. (In the modern periodic table, a group or family corresponds to one vertical column.An extended periodic table theorises about chemical elements beyond those currently known in the periodic table and proven. The periodic table allows chemists a shortcut by arranging typical elements according to their properties and putting the others into groups or families with similar chemical characteristics. Were it not for the simplification provided by this chart, students of chemistry would need to learn the properties of all 118 known elements. The term “periodic” is based on the discovery that elements show patterns in their chemical properties at certain regular intervals. Mendeleev left spaces for elements he expected to be discovered, and today’s periodic table contains 118 elements, starting with hydrogen and ending with oganesson, a chemical element first synthesized in 2002 at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia, by a team of Russian and American scientists. Its story is over 200 years old, and throughout its history, it has been a subject for debate, dispute and alteration.Īttempts to classify elements and group them in ways that explained their behavior date back to the 1700s, but the first actual periodic table is generally credited to Dmitri Ivanovich Mendeleev, a Russian chemist who in 1869 arranged 63 known elements according to their increasing atomic weight. Go into any scientist’s office or lecture hall anywhere in the world and you are likely to see one. There is no more enduring reflection of science than the Periodic Table of Chemical Elements, which sheds light not only on the essence of chemistry but physics and biology as well. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
